|
7/26/2023 0 Comments Ortho meta para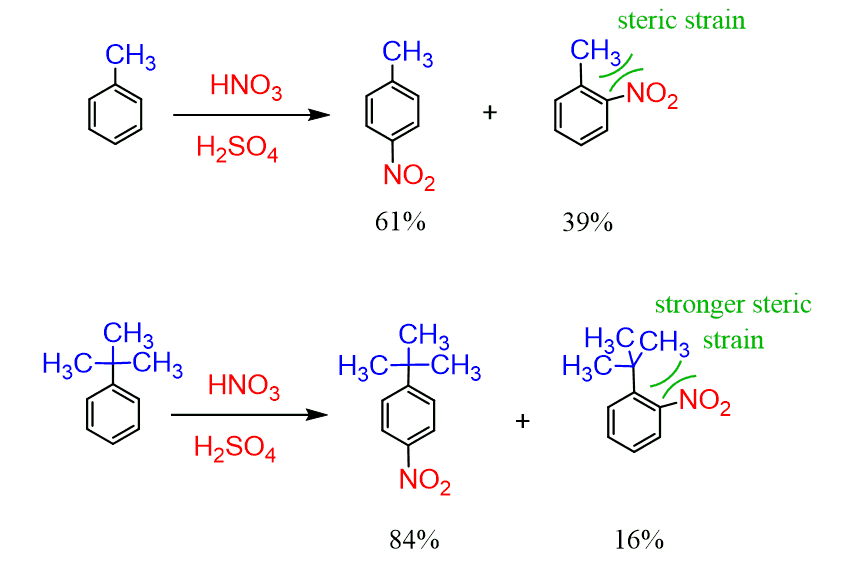
As an example for the practical applicability of the model, we performed synthetic validations via palladium-catalysed 2,2,2-trifluoroethylation reactions in our lab. For borderline cases these criteria help set the minimum reaction temperature to obtain reasonable reaction rates. we provide a simple and general protocol to estimate the reactivity of a potential substrate in C–H activation. In order to guide organic chemists, we set structural and energetic criteria to evaluate a priori the efficiency of the metalation step which is usually the rate-determining event in C–H activations, i.e. More than 250 examples have been invoked for the model, and the nature of the ortho effect was demonstrated on a wide variety of structures. We consider simple descriptors such as torsion angle and torsional energy to predict and explain the reactivity of a given substrate in directed C–H activation reactions. In this work we introduce a simple substrate-only-based model to interpret the influence of steric hindrance of a group in ortho position to the DG in directed ortho-C–H bond activation reactions, and coined the term Ortho Effect (OE) for such situations. In particular, when these groups are in neighbouring positions, their interactions can affect profoundly the efficacy of the C–H activation by transition metals.  
The success of transition metal-catalysed ortho-directed C–H activation is often plagued by the effects of undesirable interactions between the directing group (DG) and other groups introduced into the aromatic core of the substrate.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
